Metalloenzyme inhibitors
Interestingly, of the 137 NMEs, 13 target metalloenzymes, representing 7% of the drugs approved by the FDA between 2013 and 2017 DOI. Some examples that bind to the metal in the active site are shown below.
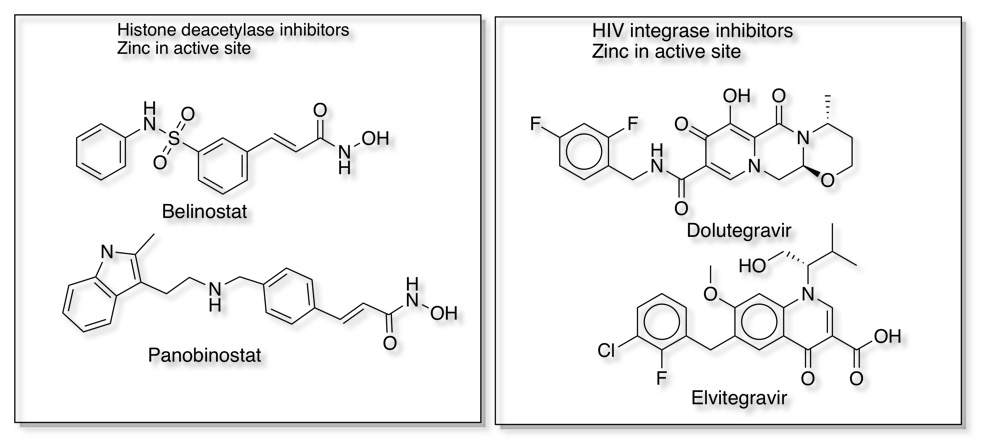
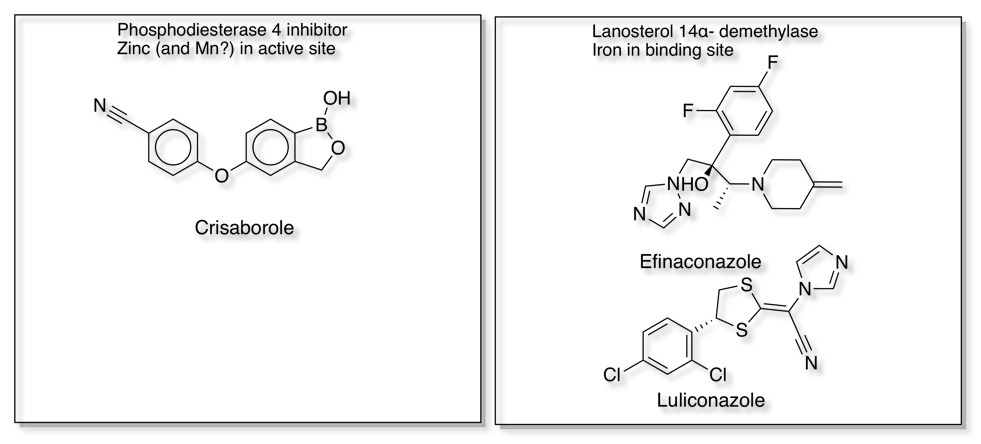
Metalloproteases
Metalloproteases are a class of enzymes that hydrolyse peptides, they all have a critical metal in the active site that is involved in the enzyme mechanism, the majority have Zinc in the active site but a few use Cobalt.
The catalytic mechanism is shown below. A water molecule activated by an active site histidine attacks the carbonyl of the scissile amide bond. The carbonyl of the amide coordinates to the Zinc to stabilise the oxyanion. The tetrahedral oxyanion then initiates elimination of the amine of the amide which then is protonated by the histidine. The protonation and deprotonation of the histidine is aided by a neighbouring acid.
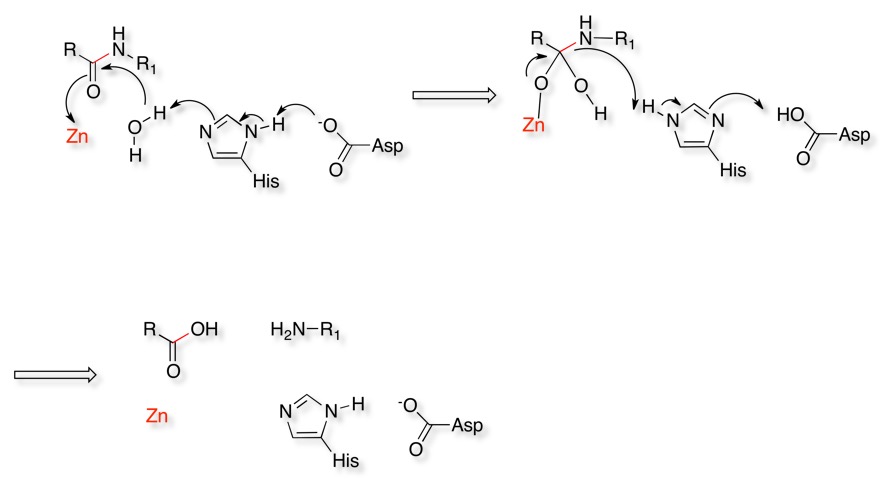
For more information on the mechanism have a look at the MACiE database (Mechanism, Annotation and Classification in Enzymes), Thermolysin, PDB entry 4TLN is an example of an Metalloprotease.
Inhibitors
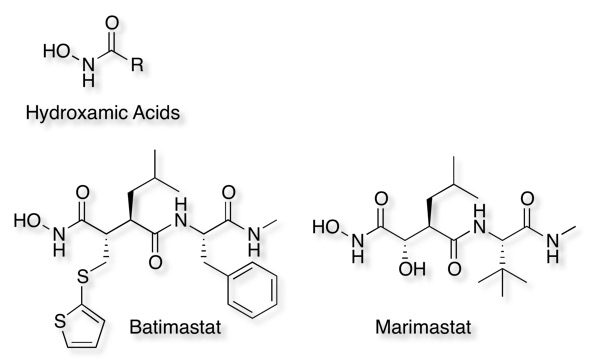
Since metalloproteases contain a metal in the active site they can be inhibited by metal chelators or by compounds that can coordinate to the Zinc. Several hydroxamic acid derivatives have been reported to be potent inhibitors and shown to act as 1,4-bidentate ligands for the zinc. A number of matrix metalloprotease inhibitors have entered clinical trials for an oncologic indication including Batimastat and Marimastat
Another metalloprotease is Angiotensin-Converting Enzyme (ACE), ACE is a Zinc-dependent dipeptidyl carboxyl metallopeptidase that converts the decapeptide Angiotensin I to the octapeptide Angiotensin II.
See also metal chelation
Useful resource MetLigDB is a publicly accessible web-based database on which the interactions between a variety of chelating groups and various central metal ions in the active site of metalloproteins can be explored in detail. Additional information can also be retrieved including protein and inhibitor names, the amino acid residues coordinated to the central metal ion, and the binding affinity of the inhibitor for the target metalloprotein.
Worth Reading
Targeting Metalloenzymes for Therapeutic Intervention DOI
Updated 2 October 2018